Abstract
Solid oxide fuel cells (SOFCs), as one of the most promising advanced energy conversion devices, has a cathode material that determines the performance of the whole cell, in which the most commonly used cathode material is Co-based chalcogenide material with excellent oxygen reduction activity and high electrical conductivity. However, Co-based chalcogenide materials are mismatched with commonly used electrolytes due to high thermal expansion coefficients (TEC), resulting in poor thermal matching between the battery cathode and electrolyte at high temperatures. In order to reduce the TEC of the cathode material, a composite cathode was prepared by adding a negative thermal expansion material to the cathode material. The prepared cathode material SmBaCo2O5+δ (SBCO) and the negative thermal expansion material Sm0.85Zn0.15MnO3 (SZMO) were mechanically mixed according to a certain mass ratio to obtain the composite cathode material (SBC-xSZM, x = 0, 5, 7.5, 10, 20 and 30 wt%), and the relevant properties of the composite cathode material were studied. The results showed that SBCO and SZMO had good chemical compatibility at 1250 °C, and the conductivity of the composites decreased significantly from 1253.27 S cm1 to 96.15 S cm1 with increasing composite amount, and the average coefficient of thermal expansion of the composites was also significantly reduced from 21.93×106 K1 (SBCO) to 17.38×106 K1 (SBC-20SZM). In addition, electrochemical impedance spectra showed that the best electrochemical performance was achieved by 5 wt% SZMO composites in SBCO cathode, corresponding to improved symmetric electrode stability.
Keywords
fuel cell; negative thermal expansion; composite cathode; conductivity; thermal expansion coefficient
1. Introduction
Solid oxide fuel cells (SOFCs) are one of the most promising advanced energy conversion technologies as they can directly convert the chemical energy of fuels into electrical and thermal energy. SOFCs are favored for their high efficiency, environmental friendliness, low operating noise, and wide range of fuel applicability [1-3]. The most commonly used cathode materials for SOFCs are Co-based chalcogenides because of their excellent oxygen reduction activity and high electrical conductivity. However, Co-based chalcogenide materials are much larger than the commonly used electrolytes LSGM (11.4×106 K1) [4-6], SDC (12.0×106 K1) [7], and GDC (13.08×106 K1) [8] due to the high thermal expansion coefficients (TEC≈20-26×106 K1)[9], which leads to poor thermo-mechanical compatibility at high temperatures, which may lead to cracking of the interface after many cycles resulting in a significant shortening of the battery life.
A lot of efforts have been made to improve the thermal match between the battery cathode and electrolyte, such as compositing the chalcogenide electrode material with the electrolyte material [10], introducing A-site defects in the chalcogenide material [11], and partially replacing the Co ions with cations such as Cu, Fe, Sc, etc. [4,12]. These methods can effectively lower the coefficient of thermal expansion, but they may also have a negative impact on the oxygen reduction reaction (ORR) of the electrode material catalytic activity negatively, so that the electrochemical performance of the optimized materials is still unsatisfactory. There is an urgent need to find a new strategy that can dramatically improve the thermal match between cathode and electrolyte while causing no or very little negative impact on the ORR catalytic activity of cathode materials. In recent years, the compounding of negative thermal expansion materials into electrode materials has become the mainstream of research [13].
In this work, the cathode material SmBaCo2O5+δ(SBCO) and the negative thermal expansion material Sm0.85Zn0.15MnO3 (SZMO) were prepared by the distribution of sol-gel method and solid phase reaction method, and the composites were obtained by mechanical composite method (SBC-xSZM, x=0, 5, 7.5, 10, 20 and 30 wt%). The crystal structure, electrical conductivity, electrochemical properties, thermal expansion coefficient and stability of the composites were investigated. The experimental results show that the composite electrodes exhibit good electrochemical properties and excellent thermal stability. The method of preparing composite electrodes using composite materials may provide an excellent and generalized strategy for developing electrodes with compatibility and high activity for SOFC.
2. Experimental Section
2.1 Material Preparation
SmBaCo2O5+δ(SBCO) cathode material [14] was prepared by sol-gel method, and the stoichiometric ratio of Sm2O3 was completely dissolved in concentrated nitric acid to form a clarified nitrate solution. An appropriate amount of deionized water was added to the nitrate solution. Then stoichiometric ratios of Ba(NO3)2 and Co(NO3)2∙6H2O were added and mixed well. The complexing agent ethylenediaminetetraacetic acid (EDTA) and citric acid (CA) were dissolved in the nitrate solution, and the molar ratio of CA : EDTA : metal ions was 1:2:1, and the pH value of the solution was adjusted to about 7 by dropping in ammonia, and the solution was diluted by adding deionized water and evaporated at 90 ℃, and then transferred to a drying box after the formation of the brown viscous gel, and the residual water in the gel was evaporated at 150 ℃. The obtained gel was then calcined at 400 ℃ for 10 h to obtain the precursor powder. Finally, the precursor powder was sintered at 1000 ℃ for 10 h to obtain the SBCO cathode powder. Sm0.85Zn0.15MnO3(SZMO) negative thermal expansion materials were prepared by solid-phase reaction method [15] using SmO2, ZnO and Mn3O2 as the raw materials, and manually milled in an onyx mortar for 48 h. The sample discs were obtained by pressing the powder into the mold at 12 MPa for 2 min. The discs were calcined in a muffle furnace at 1000 ℃ for 10 h. After cooling, the powder was continued to be manually ground in the mortar until fine powder was obtained, and finally the fine powder was calcined in a muffle furnace at 1250 ℃ for 24 h to obtain the SZMO powder.La3Sr0.9Ga0.1Mg0.8O0.2 (LSGM) electrolyte sheets were prepared by solid-phase reaction method, and the details of the preparation were consistent with that reported in the previous works. The thickness of LSGM electrolyte was polished to 300 µm for subsequent symmetric electrode and single cell assembly. The composite SBC-xSZM (x=0, 5, 7.5, 10, 20 and 30 wt%) was obtained by adding SBCO and SZMO powders proportionally to the mortar and pestle and then manually grinding for 48 h with alcohol.
2.2 Symmetrical Electrodes
The cathode material, ethyl cellulose (binder) and α-pinitol (solvent) were mixed uniformly in proportion, and a two-side symmetric square with an area of 0.16 cm2 was drawn on both sides of the electrolyte sheet with a pencil, and the mixed slurry was coated inside the square on both sides of the electrolyte sheet by the screen-printing method, and then the electrolyte sheet was put into an oven at 80 ℃ for 30 min, so as to make the slurry fixed on the two sides of the electrolyte sheet, and the slurry was fixed on both sides of the electrolyte sheet, which was repeated 2 times per side. The process was repeated twice on each side. Finally, the electrolyte sheet was calcined in a high-temperature muffle furnace at 950 ℃ for 2 h to obtain a symmetric electrode with the structure of cathode | electrolyte | cathode.
2.3 Characterization Methods
The phase structure of the materials was characterized using an X-ray diffractometer (XRD, Rigaku Ultima IV). The experiments were carried out using Cu-Kα radiation at a voltage of 40 kV, a current of 30 mA, and a scanning range of 20° to 90°. The temperature-dependent conductivity of the cathode material was tested using a four-probe tester (Model RTS-8) with a four-terminal test method over the temperature range of 200 ℃ to 850 ℃ with an interval of 25 ℃. The thermal expansion coefficients of the dense samples were measured in the range of 50-950 °C using a thermal expansion meter (NETZSCH DIL402C type) in an air atmosphere at a heating rate of 5 °C min¹. The thermal expansion coefficients of the dense samples were measured in the range of 50-950 °C using a thermal expansion meter. The electrochemical impedance of the cathode material was tested by preparing symmetric electrodes. A sinusoidal voltage with frequency variation was applied on both sides of the symmetric electrodes to analyze the relationship between the amplitude and phase of the current signals with the frequency, and the polarization impedance of the cathode material was obtained. In the stability test, the thermal stability test was carried out in an air atmosphere, and the temperature was increased from room temperature to 700 ℃ and then held for 150 h. The area specific resistance (ASR) of the symmetric electrode was recorded every 10 h. The thermal stability test was carried out in an air atmosphere with the temperature increased from room temperature to 700 ℃. The cyclic stability test was performed in an air atmosphere with the temperature increased from room temperature to 700 ℃ at a rate of 2 ℃ min1, and then decreased from 700 ℃ to room temperature at a natural cooling rate. The ASR of the symmetric electrode was tested every 700 ℃ and the cycle was repeated 10 times. The cross-section microstructure of the symmetric electrodes was characterized by scanning electron microscopy using a model SU5000 hot-field field emission scanning electron microscope manufactured by Hitachi High-Technologies Corporation, Japan, which was operated with a ZrO/W Schottky-type electron gun using a voltage of 30 kV to observe the cross-section morphology.
3. Experimental Results and Discussion
3.1 Physical Structure
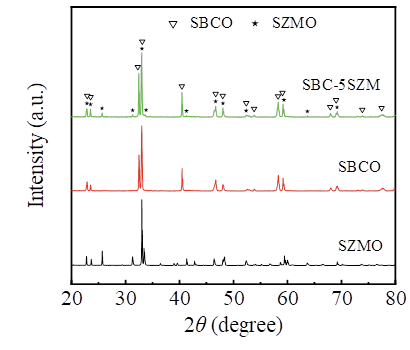
Fig. 1 demonstrates the XRD patterns of SBCO, SZMO, and SBC-5SZM composite powders. SBCO and SZMO powders were sintered at a high temperature of 1000 ℃ for 10 h. The SBCO spectral line corresponds to the SmBaCo2O5.51 (PDF#53-0133) in the standard card, which forms a double chalcocite structure [17,18] with a space group of P4/mmm (123). The SZMO spectral line corresponds to SmMnO3 (PDF#25-0747) in the standard card, forming a single chalcogenide structure [15] with a space group of Pbnm (62). The crystal structures and cellular parameters of SBCO and SZMO are listed in Table 1.SBC-5SZM composite powders were sintered at 1250 ℃ for 10 h. All of the characteristic peaks observed in the composites can be attributed to the presence of SBCO and SZMO, and no secondary phases or impurity peaks were observed in their profiles. This indicates that there is no detectable chemical reaction between SBCO and SZMO, i.e., SBCO and SZMO are still chemically compatible at 1250 ℃.
Table 1 Crystal structure, lattice parameters of SBCO and SZMO samples at room temperature
| Sample | Space group | Crystal symmetry | a(Å) | b(Å) | c(Å) | V(Å)3 |
| SBCO | P4/mmm | Orthorhombic | 3.913 | 3.913 | 7.576 | 115.29 |
| SZMO | Pbnm | Orthorhombic | 5.358 | 5.825 | 7.483 | 233.54 |
3.2 Conductivity
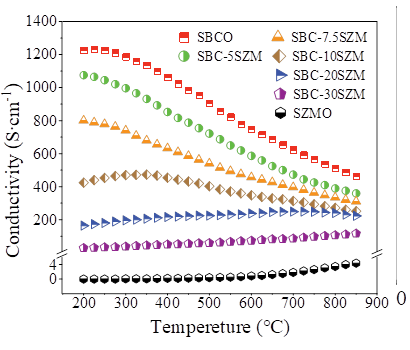
Fig. 2 shows the variation curves of the conductivity of the materials with temperature before and after the composite of SBCO and SZMO. The electronic conductivity is 2~3 orders of magnitude higher than the ionic conductivity, and the graph mainly reflects the electronic conductive properties [19]. The experimental results show that SBCO has the highest conductivity, which ranges from 1253.27 S cm1~758.23 S cm1 at 200~800 ℃. The lattice oxygen is missing at the increased temperature, and the transition metal ions undergo thermal reduction, which leads to the expansion of the lattice, which makes the electron transport distance increase and the conductivity decrease [20,21]. The conductivity of the composite SBC-xSZM (x=5~30 wt%) continues to decrease with the increase of SZMO composite ratio, which is mainly because the addition of SZMO blocks the O2 transport channel [22]. In addition, with the increase of SZMO composite ratio, the composite has a lower dependence on the temperature, which is favorable for the composites to obtain high conductivity [23] at high temperatures.
3.3 Thermal Expansion Analysis
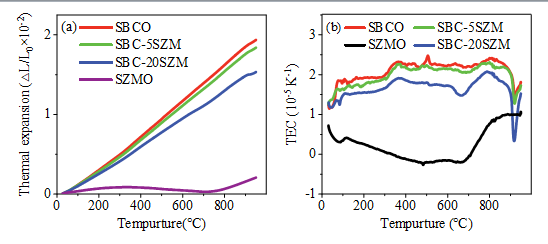
The TEC is an important index to describe the thermal stability of SOFC materials, and an excessive gap in the coefficient of thermal expansion between layers may cause cracking or even detachment between SOFC components, greatly increasing the electrode impedance and shortening the battery life [24]. As shown in Fig. 3(a), the composite with the negative thermal expansion material SZMO can effectively reduce the thermal expansion coefficient of the cathode material SBCO, and the thermal expansion coefficient is significantly reduced from 21.93×106 K1 (SBCO) to 17.38×106 K1 (SBC-20SZM). The reduction in the thermal expansion coefficient of the composite is attributed to the compensation of the thermal expansion of the negative thermal expansion material SZMO for the cathode material SBCO during the warming process. By this method, the thermo-mechanical matching between the cathode material SBC-20SZM and the electrolyte LSGM is significantly improved, which is conducive to the overall functionality and structural stability of the SOFC. Fig. 3(b) demonstrates the TEC of the composite material SBC-xSZM (x=5 and 20 wt%) before and after compositing. The TEC of the materials gradually increase at elevated temperatures, and a rapid increase of the TEC occurs at about 350 ℃, which is attributed to the transition metal undergoing thermal reduction, and the Co ions are transformed from Co4+ to Co3+ with a larger ionic radius, and at the same time, the oxygen vacancies begin to form. A sudden decrease in the coefficient of thermal expansion around 900 °C indicates a secondary phase transition, which has also been reported in PrBaCo2/3Fe2/3Cu2/3O5+δ [25] and PrBa0.9Ca0.1Co2-xZnxO5+δ [26].
3.4 Electrochemical Impedance Spectroscopy
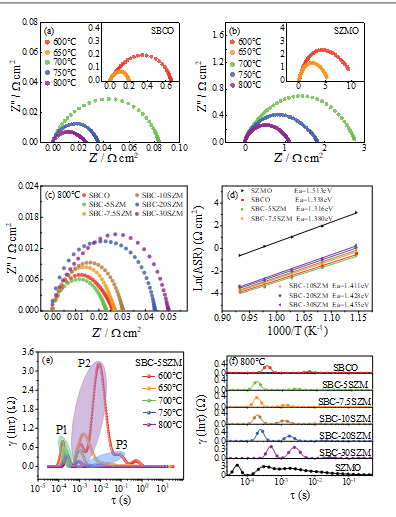
Fig. 4 EIS profiles of (a) SBCO and (b) SZMO cathodes at 600-800 °C and (c) composite cathode at 800 °C.(d) Arrhenius curve of polarization impedance versus temperature for composite and SZMO materials. (e) DRT pattern of SBC-5SZM cathode with temperature change. (f) DRT pattern of composite cathodeat 800 ℃
The electrochemical impedance properties of the materials before and after compositing are demonstrated in Fig. 4 (a-b), which show the EIS patterns of SBCO and SZMO in the range of 600-800 °C, respectively. The polarization impedance (Rp) can be determined from the intercept of the semicircular graph on the real axis [27], and it can be observed that the Rp of SZMO is about one order of magnitude higher than that of SBCO. The Rp of the material decreases continuously with increasing temperature, showing obvious thermal activation properties. Fig. 4(c) shows the EIS profiles of composite SBC-xSZM (x = 0, 5, 7.5, 10, 20 and 30 wt%) at 800 °C, and the specific values of Rp of the composites and SZMO are summarized in Table 2. The Rp of SBCO decreases and then increases with increasing the amount of SZMO composite, and the composite SBC-5SZM possesses the lowest Rp (0.023 Ω cm2), which is 11.5% lower than the Rp of SBCO (0.026 Ω cm2), and similar trends [28] were observed in other composite cathode materials. Fig. 4(d) shows the Arrhenius curves of the polarization impedance versus temperature for the composite and SZMO materials, and the polarization impedance was fitted linearly to further compute the material ORR activation energy, Ea. The results show that the SBC-5SZM possesses the lowest activation energy. Its corresponding DRT mapping is shown in Fig. 4(e)
| Sample | Rp (Ω cm2) | ||||
| 600 C | 650 C | 700 C | 750 C | 800 C | |
| SBCO | 0.661 | 0.206 | 0.084 | 0.036 | 0.026 |
| SBC-5SZM | 0.594 | 0.183 | 0.074 | 0.032 | 0.023 |
| SBC-7.5SZM | 0.831 | 0.255 | 0.091 | 0.050 | 0.027 |
| SBC-10SZM | 0.960 | 0.293 | 0.107 | 0.051 | 0.030 |
| SBC-20SZM | 1.070 | 0.331 | 0.133 | 0.073 | 0.044 |
| SBC-30SZM | 1.223 | 0.372 | 0.162 | 0.085 | 0.050 |
| SZMO | 11.451 | 5.332 | 2.756 | 1.813 | 1.109 |
The DRT plots of the composite SBC-xSZM (x = 0, 5, 7.5, 10, 20 and 30 wt%) material at the same temperature are shown in Fig. 4(e). With the increase of SZMO composite amount, the area surrounded by the DRT plot line and the horizontal axis increases continuously, and the polarization impedance increases continuously, showing the attenuated impedance performance. At the same time, the position of the characteristic peak gradually shifts to the low-frequency direction with the increase of the composite amount, which corresponds to a longer relaxation time of the ORR process and slower oxygen kinetics [29]. At a relatively low temperature, P2 dominates. After the temperature increases, the intensity of P2 peak decreases dramatically and P1 gradually dominates, indicating that the oxygen molecule dissociation process is improved while the oxygen ion conduction part gradually becomes the main part of the impedance. With the composite of SBCO and SZMO, the area of the spectral line gradually increases and the electrochemical performance of the composite gradually decreases, indicating that the excessive composite with negative thermal expansion material is not favorable to the improvement of the impedance performance of the material [30].
3.5 Stability analysis
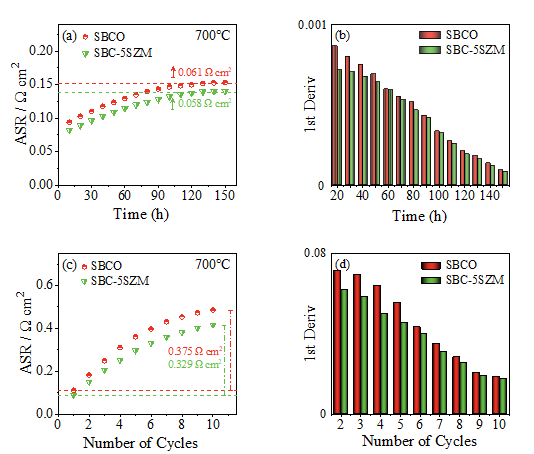
By improving the thermal match between the cathode and electrolyte materials, the cell is able to maintain excellent stability over a longer period of time. Fig. 5(a) shows the thermal stability test of the symmetric electrodes of SBCO and SBC-5SZM under air atmosphere. The ASR of the symmetric electrode of SBC-5SZM increased slowly in the first 120 h, and then stayed constant in the next 30 h, and the stabilized value of the ASR increased by 0.058 Ω cm2 compared with the initial value. The ASR of the symmetric electrode of SBCO increased rapidly in the first 130 h, and then stayed stable in the next 20 h, and the stabilized value of the ASR increased by 0.058 Ω cm2 compared with the initial value. increases and then remains constant in the next 20 h, and the stable value of ASR increases by 0.061 Ω cm2 compared with the initial value. Fig. 5(b) shows the growth rate of impedance in the thermal stability test, and the growth rate of impedance at all temperatures for SBCO is larger than that for SBC-5SZM, and the results indicate that the composite cathode material, SBC-5SZM, exhibits a more excellent thermal stability compared with the pure cathode material, SBCO. thermal stability. Fig. 5(c) shows the thermal cycling stability of the symmetric electrodes of SBCO and SBC-5SZM under air atmosphere, and the impedance performance of SBC-5SZM before and after the cathode cycling is better than that of SBCO, and the symmetric electrode impedance of SBC-5SZM increased by 0.329 Ω cm2 in the last comparison with the first one, whereas the symmetric electrode impedance of SBCO increased by 0.375 Ω cm2. Fig. 5(d) shows the growth rate of impedance in the thermal cycling test, and the growth rate of SBCO impedance at all temperatures is also larger than that of SBC-5SZM, indicating that the composite cathode, SBC-5SZM, exhibits superior electrochemical performance under the thermal cycling test compared with the pure cathode material, SBCO.
3.6 Scanning Electron Microscopy

In order to observe the effect of negative thermal expansion oxide SZMO on the contact interface between the cathode and electrolyte, scanning electron microscope (SEM) scanning was performed on the symmetric electrode cross sections of SBCO and SBC-5SZM cathodes, respectively, and a magnification of 1000 times was applied to the symmetric electrodes, and the specific experimental results are shown in Fig. 6. There is little difference in the microscopic morphology of the two different cathodes, and the cathodes are all porous structures, which are favorable for the dissociation and adsorption of oxygen at the ORR at the cathode [31]. The SEM image of the cross-section of the symmetric electrode of the SBCO cathode is shown in Fig. 6(a), and it can be clearly observed that there are some cracks at the contact interface between the cathode SBCO and the electrolyte LSGM, and these cracks substantially reduce the reactive sites at the contact surface, impede the oxygen ion transport, and increase the polarization impedance [32]. Fig. 6(b) shows the cross-sectional SEM image of the SBC-5SZM composite cathode symmetric electrode, and it can be observed that, although there are some minor cracks, the addition of SZMO drastically reduces the cracking on the contact surface between the cathode and the electrolyte, and improves the thermal matching between the two, compared to the SBCO symmetric electrode.
4. Summary
In summary, novel SBC-xSZM (x = 0, 5, 7.5, 10, 20 and 30 wt%) composite cathodes were developed and applied to LSGM electrolyte-supported solid oxide fuel cells. SBCO and SZMO showed good chemical compatibility at 1250 °C. When x increased from 0 to 30 wt%, the conductivity, on the other hand, decreased significantly from 1253.27 S cm1 to 96.15 S cm1, and the temperature dependence of the conductivity decreased gradually with the increase of x. The conductivity of SBCO-SZMO was also found to be very good at 1250 ℃. In addition, the average thermal expansion coefficient of SBC-xSZM (x = 0, 5 and 20 wt%) significantly decreased from 21.93×106 K1 (SBCO) to 17.38×106K1 (SBC-20SZM). The electrochemical impedance spectra showed that the best electrochemical performance was obtained by compounding 5 wt% SZMO in the SBCO cathode, and the impedance was reduced from 0.026 Ω cm2 to 0.023 Ω cm2. In addition, the compounding of the negative thermal expansion in the symmetric electrodes of SBCO also had a significant increase in the long-term stability and thermal cycling stability of the symmetric electrodes, and the cracking of the surface of the cathode in contact with the electrolyte was also reduced significantly, and the cathode The thermal match between the cathode and electrolyte was significantly improved. Overall, the cathode optimization method of compounding negative thermal expansion materials in the cathode is an effective strategy to improve the performance of solid oxide fuel cells.
References
- Nikiforakis, I., Sotirios, M., & Dimitris, A. (2025). Understanding solid oxide fuel cell hybridization: a critical review. Applied Energy, 377, 124277. DOI ↗ Google Scholar ↗
- Choi, S., Kucharczyk, C. J., & Liang, Y. (2018). Exceptional power density and stability at intermediate temperatures in protonic ceramic fuel cells. Nature Energy, 3(3), 202-210. DOI ↗ Google Scholar ↗
- Spreafico, C. (2025). Additive manufacturing of solid oxide fuel cells. a comprehensive review of patent literature. Journal of Power Sources, 625, 235702. DOI ↗ Google Scholar ↗
- Patra, H., Rout, S. K., & Pratihar, S. K. (2011). Thermal, electrical and electrochemical characteristics of Ba1−xSrxCo0.8Fe0.2O3−δ cathode material for intermediate temperature solid oxide fuel cells. International Journal of Hydrogen Energy, 36(18), 11904-11913. DOI ↗ Google Scholar ↗
- Anbo, Y., Tian, X., & Liping, S. (2020). Effects of rare earth doping on electrochemical properties of NdBaCo2O6-δ cathode materials. Journal of Alloys and Compounds, 837, 155563. DOI ↗ Google Scholar ↗
- Wang, S., Jin, F., & Li, L. (2017). Stability, compatibility and performance improvement of SrCo0.8Fe0.1Nb0.1O3−δ perovskite as a cathode for intermediate- temperature solid oxide fuel cells. International Journal of Hydrogen Energy, 42(7), 4465-4477. DOI ↗ Google Scholar ↗
- Zhou, Q., Wang, F., & Shen, Y. (2010). Performances of LnBaCo2O5+x-Ce0.8Sm0.2O1.9 composite cathodes for intermediate-temperature solid oxide fuel cells. Journal of Power Sources, 195(8), 2174-2181. DOI ↗ Google Scholar ↗
- Liu, Y., Shao, Z., & Mori, T. (2021). Development of nickel based cermet anode materials in solid oxide fuel cells-Now and future. Materials Reports: Energy, 1(1), 100003. DOI ↗ Google Scholar ↗
- Qiao, C., Yu, J., & Hu, S. (2024). Microstructure and electrical conductivity of La10Si6O27-La0.9Sr0.1Ga0.8Mg0.2O2.85 composite electrolytes for SOFCs. Ceramics International, 50(2), 3672-3679. DOI ↗ Google Scholar ↗
- Ding, X., Cui, C., & Guo, L. (2009). Thermal expansion and electrochemical performance of La0.7Sr0.3CuO3−δ-Sm0.2Ce0.8O2−δ composite cathode for IT- SOFCs. Journal of Alloys and Compounds, 481(1-2), 845-850. DOI ↗ Google Scholar ↗
- Zhou, W., Ran, R., (&) Shao, Z. (2008). Evaluation of A-site cation-deficient (Ba0.5Sr0.5)1−xCo0.8Fe0.2O3−δ (x> 0) perovskite as a solid-oxide fuel cell cathode. Journal of Power Sources, 182(1), 24-31. DOI ↗ Google Scholar ↗
- Zhou, W., Shao, Z., & Ran, R. (2008). Novel SrSc0.2Co0.8O3−δ as a cathode material for low temperature solid-oxide fuel cell. Electrochemistry Communications, 10(10), 1647-1651. DOI ↗ Google Scholar ↗
- Zhang, Y., Chen, B., & Guan, D. (2021). Thermal-expansion offset for high-performance fuel cell cathodes. Nature, 591(7849), 246-251. DOI ↗ Google Scholar ↗
- Wang, S., Zan, J., & Qiu, W. (2021). Evaluation of perovskite oxides LnBaCo2O5+δ (Ln= La, Pr, Nd and Sm) as cathode materials for IT-SOFC. Journal of Electroanalytical Chemistry, 886, 115144. DOI ↗ Google Scholar ↗
- Liu, X., Xi, X., & Liao, Y. & (2024). Deciphering the enhanced oxygen reduction reaction activity of PrBa0.5Sr0.5Co1.5Fe0.5O5+δ via constructing negative thermal expansion offset for high-performance solid oxide fuel cell. Applied Catalysis B: Environment and Energy, 359, 124509. DOI ↗ Google Scholar ↗
- Jiang, L., Li, A., & Duan, P. (2024). Excellence Electrochemical Performance of Fe-Doped PrBaCo2O5+δ Cathode for Intermediate-Temperature Solid Oxide Fuel Cell. Electron. Mater. Lett, 21, 579-589 DOI ↗ Google Scholar ↗
- Jun, A., Kim, J., & Shin, J. (2012). Optimization of Sr content in layered SmBa1–xSrxCo2O5+δ perovskite cathodes for intermediate-temperature solid oxide fuel cells. International Journal of Hydrogen Energy, 37(23), 18381-18388. DOI ↗ Google Scholar ↗
- Kim, J. H., Manthiram, A. (2008). LnBaCo2O5+δ oxides as cathodes for intermediate-temperature solid oxide fuel cells. Journal of the Electrochemical Society, 155(4), B385. DOI ↗ Google Scholar ↗
- Zhang, D., Zhou, J., & Luo, Y. (2023). Robust cobalt-free perovskite type electrospun nanofiber cathode for efficient electrochemical carbon dioxide reduction reaction. Journal of Power Sources, 587, 233705. DOI ↗ Google Scholar ↗
- Javed, A., Sikstrom, D., & Thangadurai, V. (2023). Investigating the electrochemical performance of Nd1-xSrxCo0.8Fe0.2O3−δ (0≤ x≤ 0.85) as cathodes for intermediate temperature solid oxide fuel cells. International Journal of Hydrogen Energy, 48(49), 18906-18917. DOI ↗ Google Scholar ↗
- Beshiwork, B. A., Wan, X., & Xu, M. (2024). A defective iron-based perovskite cathode for high-performance IT-SOFCs: Tailoring the oxygen vacancies using Nb/ Ta co-doping. Journal of Energy Chemistry, 88, 306-316. DOI ↗ Google Scholar ↗
- Lu, F., Yang, M., & Shi, Y. (2021). Application of a negative thermal expansion oxide in SOFC cathode. Ceramics International, 47(1), 1095-1100. DOI ↗ Google Scholar ↗
- Hu, L., Zhou, D., & Zhu, X. (2024). A high-performance composite cathode based on thermal expansion complementation for SOFC. Fuel, 362, 130864. DOI ↗ Google Scholar ↗
- Zheng, H., Jiang, W., & Zhang, X. (2025). Multiphysics-coupled failure mechanism of solid oxide fuel cell sealants under long-term operation: from microcrack evolution to electrochemical degradation and mechanistic damage. Composites Part B: Engineering, 301, 112527. DOI ↗ Google Scholar ↗
- Jin, F., Shen, Y., & Wang, R. (2013). Double-perovskite PrBaCo2/3Fe2/3Cu2/3O5+δ as cathode material for intermediate-temperature solid-oxide fuel cells. Journal of Power Sources, 234, 244-251. DOI ↗ Google Scholar ↗
- Liu, B., Yang, J., & Yan D. (2020). Novel PrBa0.9Ca0.1Co2-xZnxO5+δ double-perovskite as an active cathode material for high-performance proton- conducting solid oxide fuel cells. International Journal of Hydrogen Energy, 45(55), 31009-31016. DOI ↗ Google Scholar ↗
- Saccoccio, M., Jiang, C., & Gao, Y. (2017). Nb-substituted PrBaCo2O5+δ as a cathode for solid oxide fuel cells: a systematic study of structural, electrical and electrochemical properties[J]. International Journal of Hydrogen Energy, 42(30), 19204-19215. DOI ↗ Google Scholar ↗
- Jia, X., Lu, F., & Liu, K. (2022). Improved performance of IT-SOFC by negative thermal expansion Sm0.85Zn0.15MnO3 addition in Ba0.5Sr0.5Fe0.8Cu0.1Ti0.1O3-δ cathode. Journal of Physics: Condensed Matter, 34(18), 184001. DOI ↗ Google Scholar ↗
- Wang, Y., Marchetti, B., & Zhou, X. (2022). Call attention to using DRT and EIS to quantify the contributions of solid oxide cell components to the total impedance. International Journal of Hydrogen Energy, 47(83), 35437-35448. DOI ↗ Google Scholar ↗
- Chen, X., Yu, N., & Bello, I. (2023). Understanding the oxygen reduction reaction in the hierarchically oriented composite cathode with open, straight pores. Separation and Purification Technology, 325, 124713. DOI ↗ Google Scholar ↗
- Akimoto, K. (2022). Functionality of the cathode-electrolyte interlayer in protonic solid oxide fuel cells. ACS Applied Energy Materials, 5(10), 12227-12238. DOI ↗ Google Scholar ↗